Micronizzazione, cosa fa per gli integratori
Altri articoli sulla micronizzazione:
Siamo specializzati nella micronizzazione di integratori con spray dryer per prodotti ad alta biodisponibilità.
Che cosa è la micronizzazione e cosa fa?
La micronizzazione riduce la dimensione media delle particelle degli ingredienti di un integratore alla scala dei micron, impartendo nuove caratteristiche geometriche, fisiche e biologiche:
-
- aumento del rapporto superficie/volume;
- maggiore solubilità;
- aumento della biodisponibilità;
- stabilità migliorata;
- miglioramento generale delle prestazioni del prodotto.

In questo articolo esploreremo cosa fa la micronizzazione agli integratori e come lo fa.
Introduzione
La micronizzazione è un processo potente in grado di sprigionare lo straordinario potenziale terapeutico dei composti naturali contenuti negli alimenti.
Lo fa scomponendo gli ingredienti di cui sono fatti gli integratori in particelle discrete e riassemblandole in microparticelle conferendogli un nuovo incredibile potenziale terapeutico.
La micronizzazione può avvenire secondo molte tecniche tra le più rilevanti nell’industria degli integratori alimentari ci sono:
1. Jet Milling: getti ad alta velocità di gas compresso riducono le dimensioni delle particelle.
2. Ball Milling: macinazione meccanica attraverso sfere semoventi che attraverso l’impatto e l’attrito riducono la dimensione delle particelle.
3. Spray Drying: micronizza e incapsula in un solo passaggio, trasformando i liquidi in particelle fini.
4. Fluid Energy Milling: getti di gas ad alta velocità per impattare e ridurre le dimensioni delle particelle.
6. Fresatura a martelli: martelli rotanti o coltelli per abbattere le particelle attraverso l’impatto.
7. Macinazione criogenica: temperature estremamente basse per rendere fragili i materiali seguiti dalla macinazione.
8. Fresatura ad ultrasuoni: onde sonore ad alta frequenza inducono cavitazione e riduzione delle dimensioni.
9. Bead Milling: macinazione in una sospensione liquida contenente perle solide che riducono le dimensioni attraverso forze di taglio e impatto.
10. Nanomilling: una forma specializzata di macinazione utilizzata per produrre nanoparticelle con dimensioni e distribuzione precise.
La scienza dietro la micronizzazione degli integratori
I principi tecnici alla base delle varie tecniche di micronizzazione sono molto variegati, ma nonostante ciò tutte le tecniche operano aumentano il rapporto superficie/volume (SA:Vr), che è il principale responsabile dei vantaggi caratteristici della micronizzazione, ovvero aumento della:
- Biodisponibilità
- Solubilità
- Fluidità
- Miscibilità
- Stabilità

Il rapporto SA:Vr è la misura dell’area superficiale per unità di volume e si calcola dividendo la superficie della microparticella per il suo volume. Quando si riducono le dimensioni, le particelle risultanti avranno un SA:Vr più alto rispetto alle particelle dell’ingrediente non micronizzato. Ciò è dovuto alla geometria della sfera. Le particelle, come è ovvio, possono avere diverse forme, ma per comodità è convenzione immaginarle come sfere. Visto che l’area della superficie di una sfera è proporzionale al quadrato del suo raggio, mentre il suo volume è proporzionale al cubo del suo raggio, significa che diminuendo le dimensioni di una sfera, ovvero diminuendone il raggio, l’area della superficie diminuisce meno del volume, risultando in particelle con una superficie maggiore per unità di volume.

Per rendere meglio l’idea di come l’aumento del rapporto superficie/volume (SA:Vr) influisce sull’efficacia immagina una particella di uno degli ingredienti di un integratore. Questa è composta da molecole identiche tenute insieme da interazioni fisiche (forze di van der Waals). Quando sommersa in acqua questa è circondata da molecole di acqua che iniziano a scontrarsi con la superficie della particella. Durante queste collisioni, le molecole di acqua interagiscono con le molecole dell’ingrediente sulla superficie della particella, più frequenti sono queste collisioni maggiore è la velocità con cui l’ingrediente si dissolve. E ovviamente il numero di collisioni aumenta con la superficie della particella. Maggiore è la superficie risetto al volume (ovvero maggiore è SA:Vr) più velocemente l’ingrediente si dissolve.

Spesso gli integratori sono formulati in modo da racchiudere gli ingredienti attivi all’interno di una matrice. Pertanto, prima che inizi la dissoluzione, deve prima avvenire un altro processo: la liberazione, ovvero l’uscita dell’ingrediente attivo dalla matrice. Questo introduce un altro concetto: la velocità di liberazione, che si riferisce alla velocità con cui le molecole di attivo vengono rilasciate dalla forma di dosaggio diventando disponibili per la dissoluzione in acqua. Anche la liberazione inizia all’interfaccia matrice-acqua, e anche in questo caso la velocità di liberazione è governata dal rapporto superficie volume della particella, maggiore è tale rapporto maggiore è la velocità di liberazione.
Sia la velocità di liberazione che quella di dissoluzione influenzano direttamente la solubilità, l’assorbimento e l’efficacia di un integratore.

Quando somministrati per via orale, gli integratori si fanno strada attraverso il tubo digerente, attraversando prima lo stomaco e i suoi succhi gastrici (pH<2) per poi venire assorbiti a livello della membrana enterica (pH>6). Questa è ricoperte da uno strato di muco acquoso chiamato glicocalice, in cui gli ingredienti attivi si devono disciogliere prima di entrare in contatto con la membrana ed essere assorbiti.

Quando l’integratore è fatto di ingredienti attivi poco solubili in acqua (Vitamina A: retinolo, beta-carotene, Vitamina D: ergocalciferolo (D2), colecalciferolo (D3), Vitamina E: tocoferolo, Vitamina F: acidi grassi essenziali, come l’acido linoleico e l’acido alfa-linolenico, Vitamina K:) il numero di molecole attive che si si scioglieranno nel glicocalice sarà molto basso. Anche l’assorbimento sarà basso così come la loro concentrazione ematica (biodisponibilità). In questo scenario la maggior parte delle molecole dell’ingrediente rimangono intrappolate all’interno della matrice della forma di dosaggio, e infine espulsa. L’intero potenziale terapeutico verrà sprecato, e chi usa questo integratore non beneficerà degli effetti fisiologici dei suoi ingredienti attivi e probabilmente cambierà prodotto.
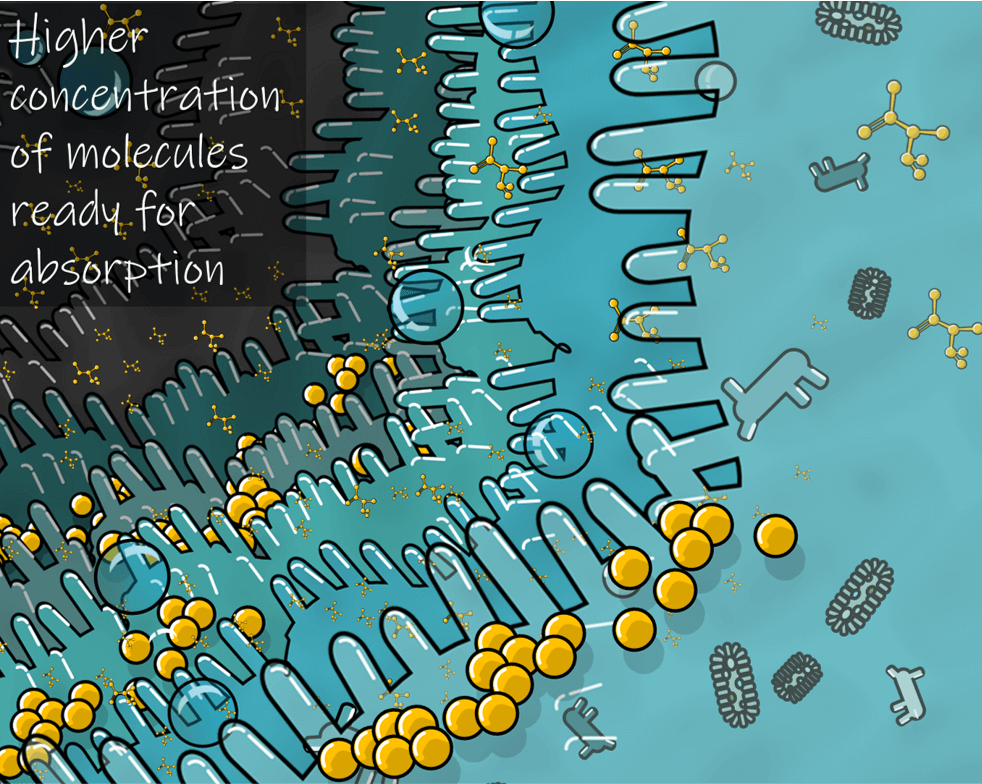
Immaginiamo ora come un integratore micronizzato possa influire sui benefici fisiologici di chi lo usa. Come il prodotto precedente questo arriverà al glicocalice e anche se i suoi ingredienti attivi restano poco solubili in acqua questi saranno sotto forma di microparticelle con un elevato rapporto superficie/volume. Ovvero il numero di molecole di ingredienti attivi capaci di dissolversi nel glicocalice è molto superiore, e questo ovviamente aumenta l’assorbimento e la biodisponibilità. L’integratore micronizzato sarà capace di sprigionare tutto il suo potenziale terapeutico e chi lo ha usato proverà un sostanziale miglioramento del proprio benessere e continuerà a comprare l’integratore.
Altri vantaggi
I vantaggi della micronizzazione non si limitano soltanto al miglioramento della solubilità in acqua ma hanno risvolti positivi anche sull’efficienza di produzione e la stabilità dell’integratore. Ecco alcuni esempi:
Dispersibilità
Si riferisce alla capacità di una polvere di essere distribuita uniformemente quando in una miscela con altre sostanze, con conseguente maggiore omogeneità della miscela, importante durante la produzione di integratori con più di un ingrediente.
Flessibilità di produzione
Grazie alla maggiore omogeneità della miscela, è possibile una facile segmentazione di linea in una più ampia varietà di forme di dosaggio come: bustine, granulati, compresse, capsule, sospensioni ed emulsioni.
Sospensioni ed emulsioni
Le particelle micronizzate hanno meno probabilità di precipitare, aumentando la stabilità di questi prodotti.
Stabilità
La micronizzazione allunga la data di scadenza dei prodotti. La migliorata omogeneità influisce positivamente anche sulla stabilità dei prodotti micronizzati, riducendo il rischio di segregazione e garantendo una distribuzione uniforme nella forma di dosaggio i micronizzati mostrano una ridotta vulnerabilità ai processi di degradazione, prolungando così la data di scadenza.
Casi studio
La micronizzazione si è dimostrata efficace sul coenzima Q10, sull’estratto di semi d’uva e sulla L-arginina ed è stato riscontrato che la loro stabilità è significativamente migliorata poiché il rischio di segregazione delle particelle è ridotto al minimo. Gli autori discutono anche dei potenziali vantaggi per l’industria degli integratori alimentari, suggerendo come la micronizzazione potrebbe migliorare la stabilità di un’ampia gamma di principi attivi, tra cui vitamine, minerali e prodotti botanici.
Micronization of Active Ingredients in Dietary Supplements: Impact on Stability and Bioavailability. Journal of Functional Foods, 10, 75-84.
Spray Drying: una tecnica di micronizzazione unica
Lo spray drying è un processo di micronizzazione che va oltre la semplice riduzione delle dimensioni, in un solo passaggio è in grado di micronizzare e incapsulare gli ingredienti di un integratore all’interno di una microparticella. Il processo di spray drying rende possibile l’inclusione di adiuvanti che impartiscono caratteristiche intelligenti alle microparticelle, come il rilascio controllato pH dipendente. In questo caso si parla di micro sistemi di rilascio capaci di massimizzare l’efficacia dell’integratore.
Ecco come funziona: i sistemi di rilascio controllato liberano i composti attivi lentamente, mantenendo più a lungo i livelli ematici all’interno della finestra terapeutica e migliorandone l’efficacia. Il sistema di rilascio può anche essere progettato per essere intelligente e utilizzare il pH per riconoscere la sua posizione all’interno del tratto gastro-enterico comportandosi di conseguenza: trattenendo i composti attivi a pH inferiori a 2 (nello stomaco) e rilasciandoli quando il pH è superiore a 6 (nell’intestino). Così, prevenendo l’inutile rilascio gastrico e concentrando il carico terapeutico nel sito di assorbimento, ottenendo i seguenti vantaggi:
Protezione: il composto attivo è preservato dalla degradazione nello stomaco.
Maggiore assorbimento: del principio attivo naturale attraverso la parete intestinale.
Maggiore efficacia: il rilascio controllato mantiene costanti i livelli ematici entro le finestre terapeutiche aumentando l’efficacia.
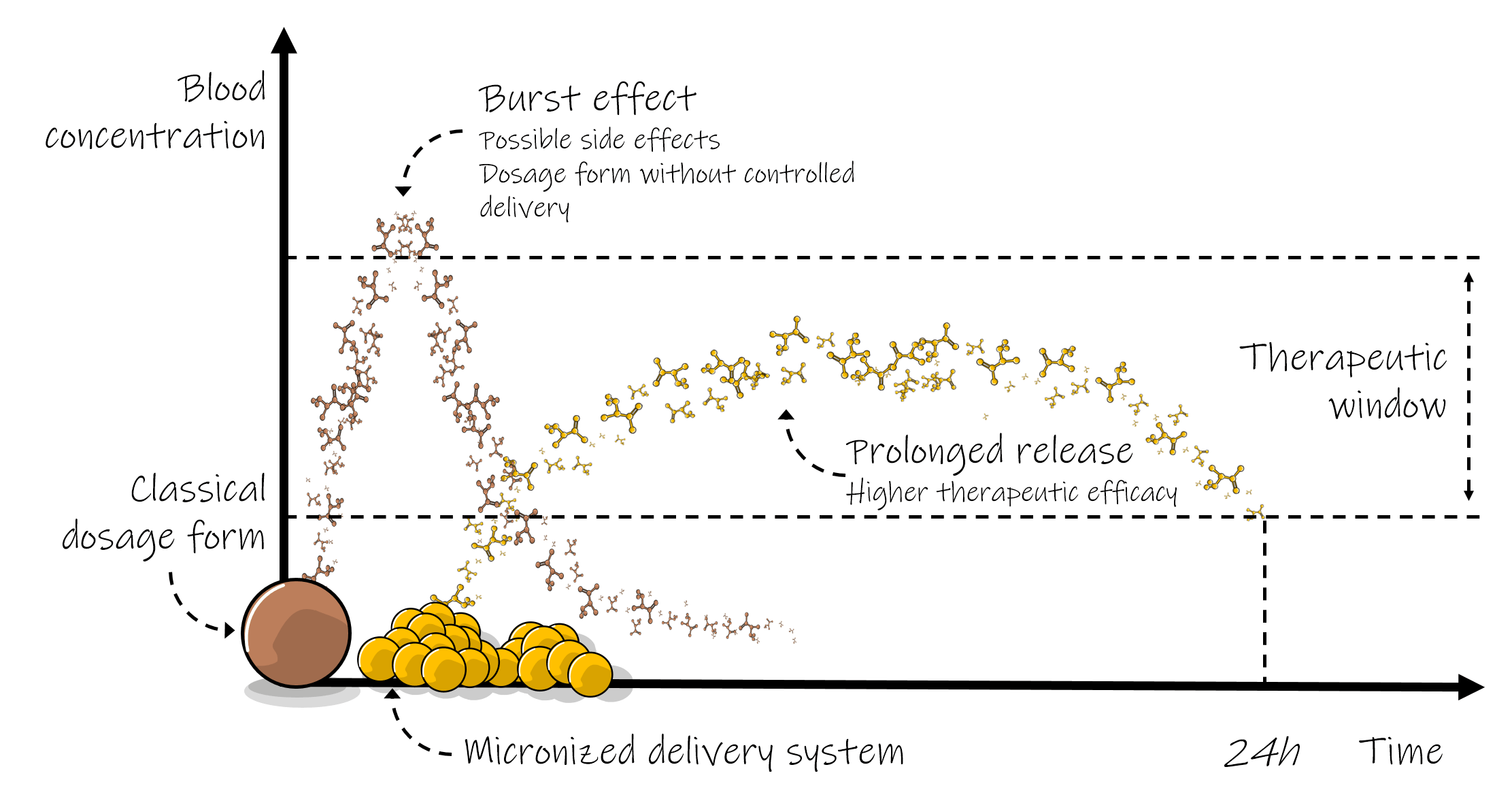
Technology Scientific è la SME nata spin-off La nostra capacità innovativa serve le migliori aziende e startup






GUIDA PDF GRATUITA
La guida completa alla
progettazione di processi di micronizzazione di integratori
La guida definitiva e gratuita per progettare,
realizzare e lanciare il tuo integratore micronizzato.
Siamo specializzati nella micronizzazione di integratori e nutraceutici con spray dryer per una maggiore biodisponibilità.
Sviluppo
Prototipazione su scala di laboratorio
Studi di preformulazione
Sviluppo della formula
Convalida dell’efficacia
Industrializzazione
Scale-up
Produzione pilota conforme a GMP
Shelf-life secondo le linee guida ICH
Produzione
Produzione GMP
Siamo specialisti dello spray drying per prodotti salutistici, integratori e nutraceutici. Ottimizziamo la biodisponibilità progettando microparticelle amorfe, stabili e con un elevato rapporto superficie/volume.
Technology Scientific è la SME nata spin-off La nostra capacità innovativa serve le migliori aziende e startup






Con le micronizzazioni di Technology Scientific,

